This remarkably fast approval turnaround for a medical device of this complexity has been partly attributed to the granting of a Breakthrough Device Designation by the US Food and Drug Administration (FDA) in 2022, and the impact of “company-changing funding” provided by MTPConnect’s Clinical Translation and Commercialisation Medtech (CTCM) program.
Tens of thousands of central venous catheters are inserted into the veins of newborns and children in Australian Intensive Care Units every year to deliver fluids and administer life-saving medications and nutrients. Current techniques can be hit and miss when it comes to correct positioning of the catheters.
“Clinicians are really trying their best, but the current system is difficult,” said co-founder and Chief Operating Officer Shing Yue Sheung. “Around 40 per cent of the time, the catheter tip is placed in the wrong spot.”
The consequences of misplaced catheters can be severe, potentially causing tissue damage if infusions are delivered to the wrong area. Catheter migration once inserted is also another concern.
Navi’s groundbreaking device uses techniques that have been proven in adult treatment but never in neonatal care.
“For the correct positioning of a catheter in an adult procedure, we’re talking about a ‘landing zone’ of two to three centimetres,” said Shing.
“But when the procedure is for a sick baby, that is only around 0.5 centimetres, with much greater consequences for a misplaced catheter. The way that we have designed our system is very accurate for newborn patients.”
The catheters themselves are tiny – a third of a millimetre in diameter, little bigger than a human hair.
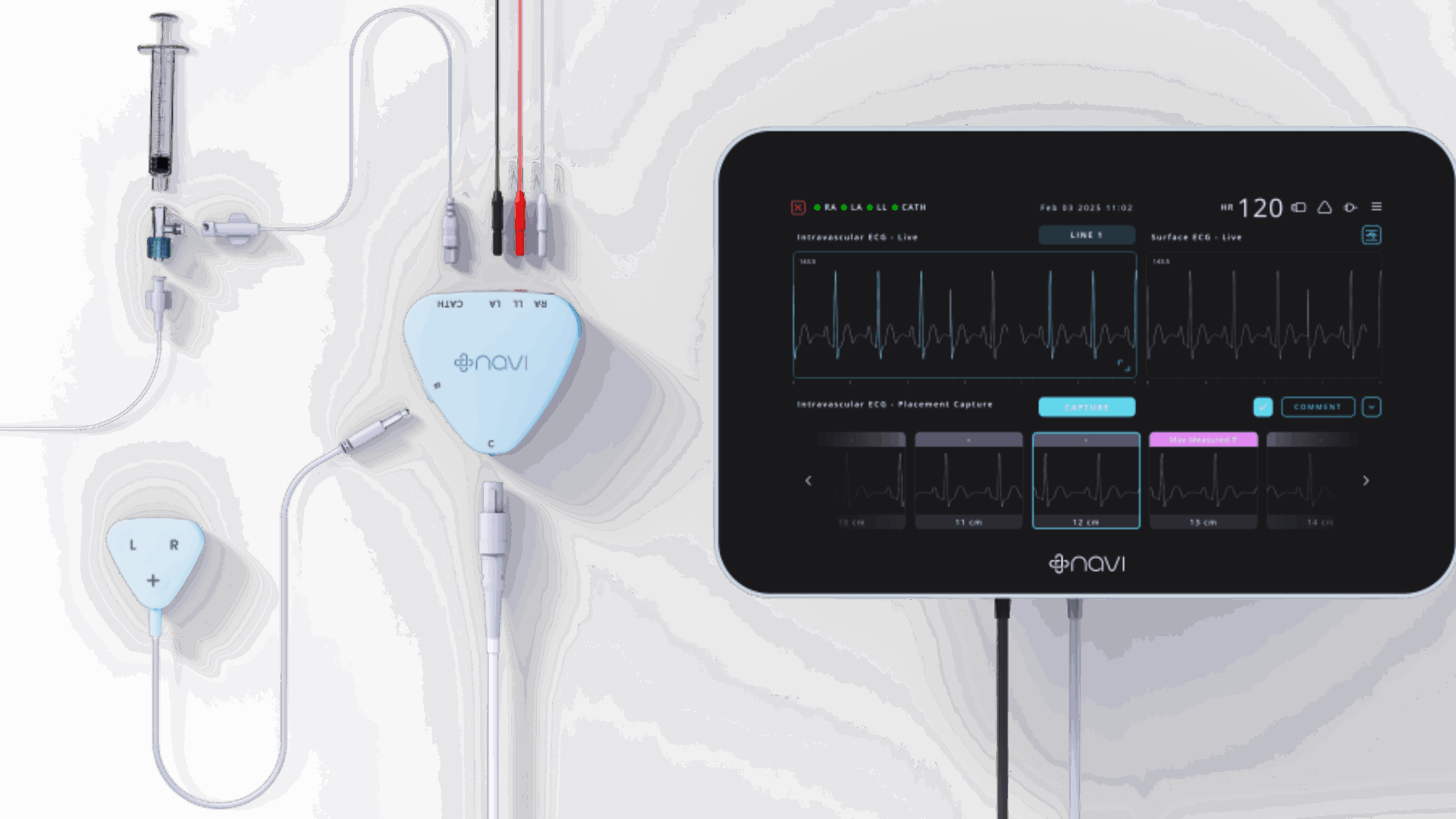
“It’s very thin, it’s extremely flexible, and even the slightest misplacement can cause some complications,” said Shing. “Placement is based on a blind insertion and the current standard of care is to use X-ray, which is post-procedure, exposes patients to radiation and causes a lot of frustration to the clinicians because they’re waiting for an X-ray to be called over and they can’t confirm the tip position until the X-ray is performed.
“Sometimes it can take a long time for an X-ray to be taken – maybe 30 to 45 minutes – especially in the middle of the night,” said Shing.
Navi’s device uses sensors and the patient’s own heart signals. As the catheter gets closer to the source of those electrical impulses there is a specific change in the heart signal.
The information is used to provide real-time feedback to clinicians on the location of the catheter tip, so they know exactly where the catheter is located, both during and after the placement procedure.
“We display this information on a screen and clinicians can make any kind of adjustments in real time,” said Shing.
Navi began developing the new device in 2018 and credits strategic decisions, strong clinical demand, and critical funding and support from the CTCM program as key drivers of this accelerated timeline.
“MTPConnect’s funding was company-changing for us,” Shing said. “It allowed us to complete essential studies, finalise our device design, grow our team, and even start manufacturing – all in just two years.
“MTPConnect’s connections and advice on regulatory, manufacturing, and commercial strategy were invaluable.”
During the CTCM-funded project the Navi team progressed the device from Technology Readiness Level (TRL) 5 to TRL 7 over 24 months. Through the CTCM program they were matched with Cicada Innovations as a program partner to assist with this challenge by providing strategic guidance.

The team powering Navi Medical Technologies
The company has been able to refine the device’s design with extensive clinician and end-user engagement to ensure optimal performance in clinical settings. Navi worked closely with neonatal and paediatric healthcare professionals conducting 200 clinical interviews across Australia and the US and conducting more than 100 product demonstrations to build product awareness among healthcare professionals.
These interviews provided valuable feedback. The Neonav® underwent six device iterations, requiring a dedicated multi-disciplinary team that expanded from 8 to 12 staff making improvements based on feedback from users and data collected during testing and studies. Each iteration allowed the team to resolve technical issues, enhance the user experience, and improve the accuracy of catheter tip placement detection.
Collaborating with the University of Melbourne, and the Royal Women’s Hospital as well as D+I and Entech helped to foster a successful culture of innovation, making sure the technology could eventually be adapted for use in various healthcare systems around the world to improve neonatal care.
Not only does Neonav® make clinicians work more efficient, and less stressful, it reduces risks of complications, leading to improved health outcomes and potentially shorter hospital stays for these young and vulnerable patients.
This also translates to less stress for families and lower healthcare costs.
The Neonav® device has the potential to enable more hospitals to offer effective specialist care for critically ill children, especially for families requiring neonatal intensive care.
The device is also open source in that, unlike many of the adult devices, it is not tied to one brand of catheter.
“We try to be agnostic to all brands of catheters so that hospitals can choose their own, which is actually something that the users have asked us to do,” Shing said.
For the Navi team who came together originally from The University of Melbourne and Melbourne Business School’s Biodesign Innovation Subject to work with senior neonatologist Associate Professor Christiane Theda, this exciting journey is about to take on a new dimension.
With FDA clearance in hand, the company is now gearing up for its first commercial launch in the US, aiming to get the device in hospitals as soon as possible. This breakthrough technology promises to transform the standard of care for some of the most vulnerable patients in neonatal and paediatric intensive care.
Since initiating their CTCM project, the company has won the Victorian Premier’s Design Award – “Best Product Design” (2024) for their device and attracted significant further investment of more than $3 million including funding from the Victorian Government’s Breakthrough Victoria Fund, a CRC-P Round 14 Grant (2023), a City of Melbourne Small Business Grant (2023), the Victorian Government’s Medtech Manufacturing Growth Program Grant (2024) and the FDA funded Midwest Pediatric Device Consortium Fall Grant (2024).
Exciting times for a Melbourne startup with big ambitions to create brighter, healthier futures for children.
https://www.mtpconnect.org.au/Image?Action=View&Image_id=6683