
AF occurs when the heart’s electrical network become unsynchronised, resulting in an irregular heartbeat. This can trigger a range of distressing symptoms, including palpitations, chest discomfort, fatigue, dizziness and shortness of breath, all of which can significantly disrupt daily life and emotional wellbeing. In severe cases, AF can lead to serious complications such as blood clots, stroke, heart failure, dementia or even death. The condition also imposes a financial burden, driven by hospital stays and expensive treatment options
Catheter ablation is a common treatment for AF, particularly for a proportion of patients whose symptoms are inadequately controlled by medication, with success rates ranging between 50 and 70 per cent[4]. The process involves inserting a small catheter into the heart to target and destroy cardiac tissue, typically using thermal or cryo energy. Although complications are relatively rare, they can be severe, with an early mortality rate of 1 in 200 patients within the initial admission or 30-day readmission[5]. The procedure can also cause collateral damage to the healthy tissue and structures surrounding the target. Due to the risks involved, catheter ablation is typically only performed by highly skilled specialists using expensive, complex catheter devices. Repeat procedures are often necessary, further driving up costs and resource use.
Established in 1999, New South Wales-based company CathRx is Australia’s only onshore designer and manufacturer of electrophysiology catheters. It is developing an innovative device to make catheter ablation safer, faster and more effective, setting a new standard for this life-changing procedure.
Revolutionising catheter ablation with dual-function technology
CathRx operates with a robust Quality Management System and manufacturing facility which are ISO13485 and MDSAP-certified. The company manufactures a range of diagnostic catheters (KhelixTM) with a modular design, that gives clinicians the flexibility to adapt them to match the procedure, the patient’s anatomy and their own preferences.
CathRx has pioneered the development of reusable electrophysiology catheters, which have demonstrated the ability to reduce medical waste by up to 70 per cent compared to conventional single-use systems. CathRx’s technology is also designed to be more affordable, saving the healthcare system up to 50 per cent per catheter use.
The company has built strong commercial relationships with global distributors, and its products are already available or approved in major markets such as the US, Europe, South Korea and Australia.
Leveraging the team’s expertise, CathRx is developing ElectroPulseTM, a non-thermal, tissue-selective pulsed electric field ablation (PFA) System. In contrast to thermal or cryo ablation procedures, which are non tissue selective, the ElectroPulse utilises short microsecond bursts of high-amplitude electrical pulses that ablate or destroy cardiac tissue while preserving surrounding tissue. The catheter is also designed to be one of the smallest catheters in its class for better handling and safety, and dual-function, combining diagnostic capabilities with therapeutic PFA delivery.
Backing ElectroPulse Technology
CathRx was awarded $1.5 million funding from MTPConnect’s Clinical Translation and Commercialisation Medtech (CTCM) accelerator program in 2023 to progress its innovation from Technology Readiness Level (TRL) 5 to TRL 7. This included support and mentorship from program partner, the Medical Technology Association of Australia (MTAA). The project spanned the small-scale manufacture of CathRx’s trial device and design improvements, as well as the completion of its first-in-human clinical study.
This all-in-one solution enables clinicians to perform real-time mapping and ablation seamlessly, enhancing patient safety, with no disruption to existing clinical workflows, and reducing costs. Combining both functions into a single device eliminates the need for a separate mapping catheter, resulting in a simpler quicker procedure and approximate savings of $2,300 per procedure in equipment costs.
A simple and faster procedure also allows for same day discharge significantly improving patient outcomes and reducing cost to healthcare system. There’s also the potential for additional savings from fewer hospital admissions and repeat procedures.

The ElectroPulse Variable Loop Ablation Catheter
Cross-sector collaboration delivers clinical validation
CathRx took a highly collaborative approach to this project, bringing industry, clinical and research sectors together in a highly integrated way. The clinical trial was conducted in partnership with the Centre for Heart Rhythm Disorders (CHRD) and University of Adelaide. Additionally, a steering committee comprising top electrophysiologists from 6 hospitals across Australia and New Zealand provided strategic oversight. This cross-sector collaboration exemplified a progressive way of working in the Australian medtech ecosystem, combining sovereign manufacturing capability with world-class clinical research.
The first-in-human study successfully achieved its primary objectives, including demonstrating acute procedural success, confirming safety in human subjects, and laying the foundation for long-term efficacy.
Over the course of the program, 75 patients were treated with the ElectroPulse system, and a comprehensive clinical trial report was produced, confirming long-term procedural success. The trial’s initial results have been published and showcased at prominent conferences, including the Heart Rhythm Society’s annual meeting, earning widespread peer recognition for the project.
The device likewise garnered positive feedback from clinicians, who are the intended users of the device.
“The ElectroPulse PFA system has performed exceptionally well in our clinical experience,” said Professor Prash Sanders, Director of the CHRD in Adelaide and member of CathRx Medical Advisory Board.
“It’s intuitive to use, delivers consistent results, and has shown a strong safety profile with promising efficacy. As a second-generation product, it brings meaningful improvements and is well-positioned to compete in the global market.”
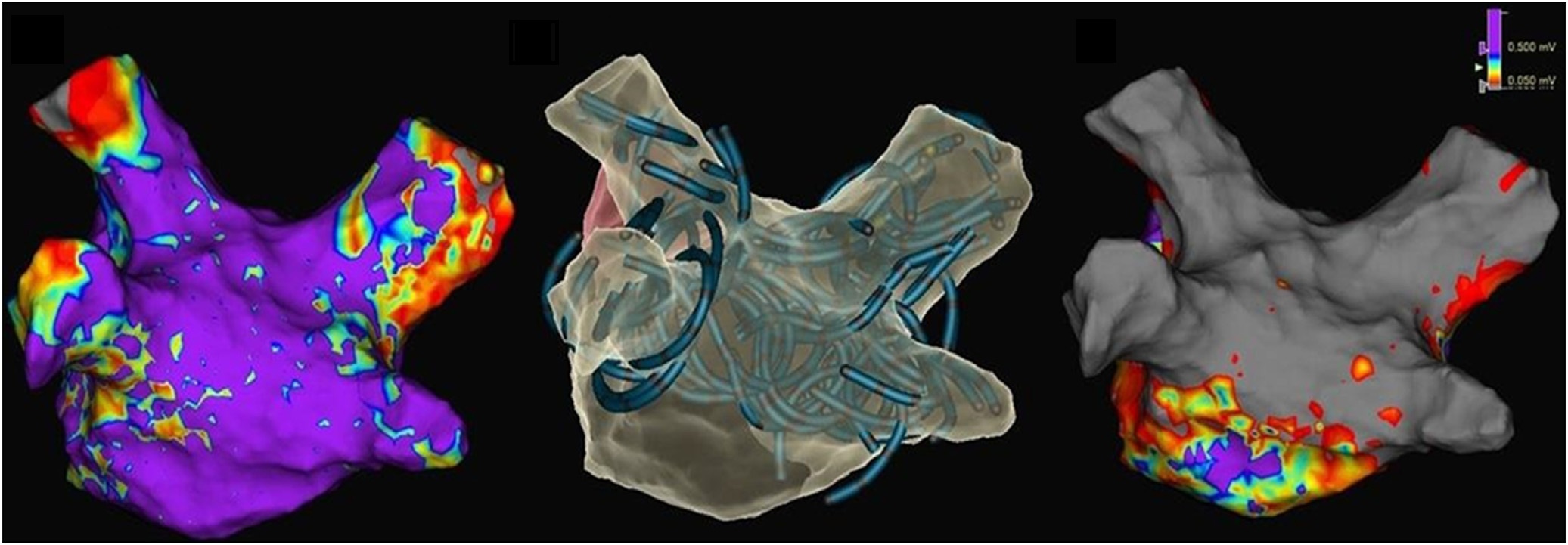
3D electro anatomical maps – pre ablation (left), post ablation (right), catheter placement shadows (middle)
CTCM support accelerates journey towards global commercialisation
The CTCM program played a pivotal role in enhancing CathRx’s manufacturing capabilities and research infrastructure. By facilitating the purchase of a laser cutter, the program ensured seamless continuity in catheter production. Additionally, upgrades to the company’s IT infrastructure have further strengthened operations. These advancements provide a solid foundation for future scalability and drive CathRx’s journey towards digital transformation.
CathRx CEO Ian Fong said: “Support from MTPConnect through the CTCM grant has been instrumental for CathRx. It enabled us to upgrade essential infrastructure and fast-track the development of our ElectroPulse PFA system. We’ve successfully demonstrated the safety and effectiveness of the system in a large first-in-human study – something rarely achieved at this scale without dedicated funding like that provided through the CTCM program.”
CathRx is making significant strides towards bringing the ElectroPulse system into the hands of more clinicians and electrophysiologists. Through its CTCM project, the company has developed invaluable in-house expertise in conducting clinical trials, paving the way for the next phase: multi-centre clinical trials. These trials will generate robust clinical data to support regulatory submissions.
CathRx is fully prepared and eager to take this next step, having secured ethics approval for the multi-centre trial and recently receiving new funding from MTPConnect’s Targeted Translation Research Accelerator (TTRA) Drugs and Devices program.
“Over the next year, we expect strong results from our multi-centre clinical trial, which will be a major step towards global commercialisation,” said Ian Fong.
The company has also expanded its manufacturing capabilities through a partnership with a contract manufacturer in Taiwan, allowing for increased product supply once regulatory approval is obtained. CathRx is also actively pursuing new distribution partners in regions beyond its current reach, such as Latin America and South-East Asia, while remaining open to strategic investors that can support its global expansion efforts.
The ElectroPulse PFA system represents a promising breakthrough in the treatment of AF, offering a safer, faster and more effective solution. Backed by robust clinical data and growing interest from international distributors, CathRx is well-positioned to bring this innovation to market.
[1] https://www.thelancet.com/journals/lanepe/article/PIIS2666-7762(23)00205-3/fulltext
[2] https://www.sciencedirect.com/science/article/pii/S1098301524000354
[3] https://www.aihw.gov.au/reports/heart-stroke-vascular-diseases/hsvd-facts/contents/all-heart-stroke-and-vascular-disease/atrial-fibrillation
[4] https://pmc.ncbi.nlm.nih.gov/articles/PMC5135164/
[5] https://www.jacc.org/doi/10.1016/j.jacc.2019.08.1036