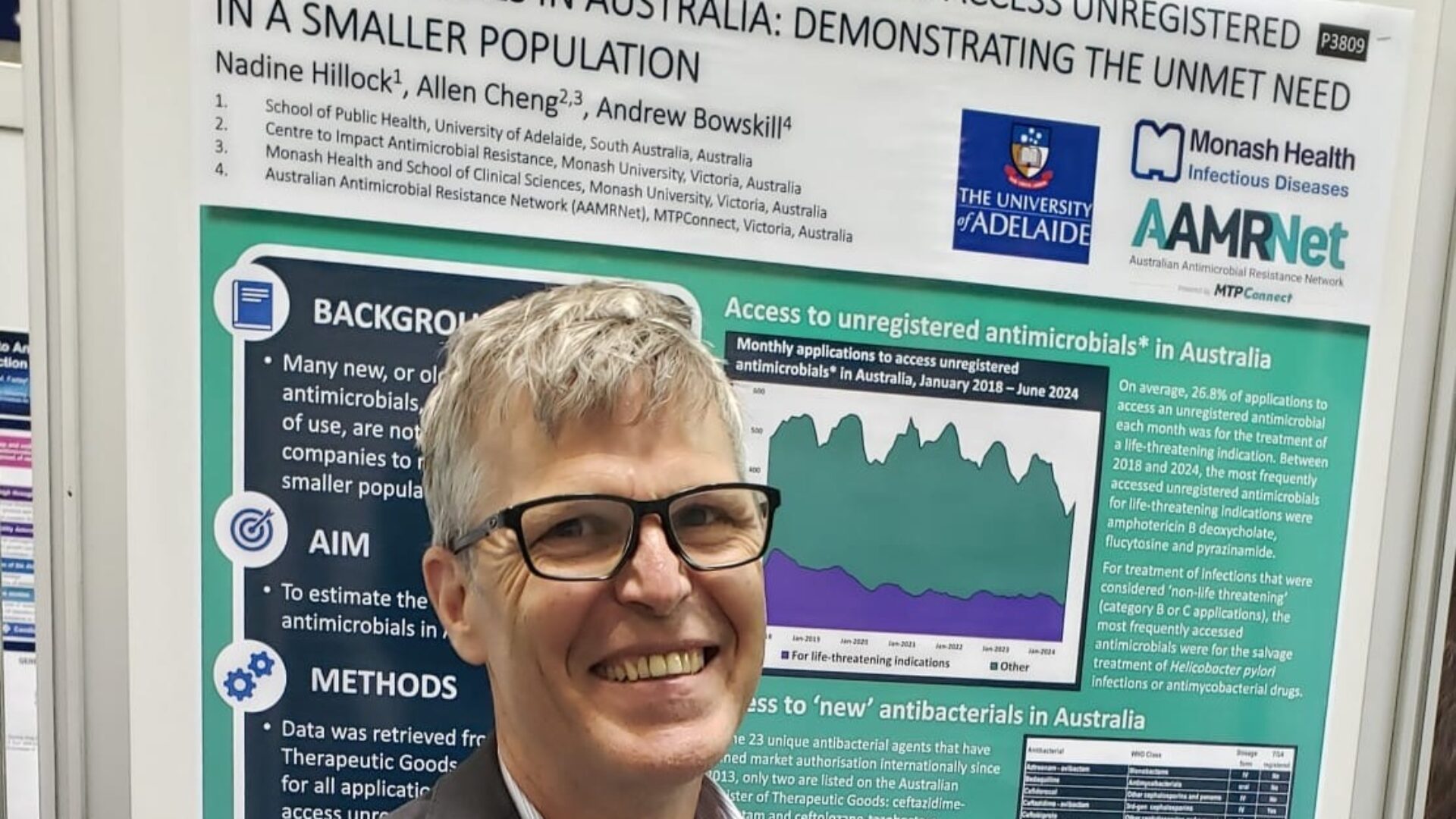
AAMRNet has been participating in Congress activities, and as co-chair I have been presenting a research poster, co-authored with Nadine Hillock from the University of Adelaide and Allen Cheng from Monash University, to highlight the concerning trends in prescriber applications to access unregistered antimicrobials in Australia: demonstrating the unmet need in a smaller population.
On the eve of the Congress, CARB-X held their third annual Investor Day, bringing together investors with CARB-X Product Developers whose programs are maturing towards or into human clinical trials.
Buzz from CARB-X Investor Day
CARB-X is the world’s leading provider of non-dilutive funding for the acceleration of antibacterial products, making its portfolio the best in the world for early-stage innovations targeting the most critical pathogens. The Investor Day gives investors with an interest in the space a chance to hear about the best early-stage opportunities in the world.
As we know, the R&D and commercialisation of innovations to prevent, diagnose and treat bacterial infection is particularly challenging, though it is not without opportunity. This was well demonstrated earlier this year when BioVersys, a company formerly funded by CARB-X, completed a highly successful IPO on the Swiss exchange, valuing the company at CHF 216 million.
Around 20 companies from all over the globe presented throughout the day, representing the world’s best AMR targeted early-stage innovations in diagnostics, vaccines and other preventatives, and Direct-acting and Non-traditional Therapeutics. Each company had 15 minutes to present and take questions, showcasing many different approaches to tackling AMR, including phage therapies, vaccines, anti-biofilm agents, and small and large molecule therapeutics with novel mechanisms of action.
It was interesting to see that several of the companies have diversified their pipelines, developing therapies in other areas such as inflammation.
The importance of global collaboration to progress projects through to commercialisation was highlighted, presenting a strong opportunity for Australia with its highly regarded clinical trials landscape.
Interest in Australia’s clinical trials ecosystem is certainly strong, with several companies expressing an interest in conducting phase I trials in Australia, and take advantage of the fast startup enabled by the Clinical Trials Notification (CTN) scheme. And the interest and opportunity goes beyond phase I trials, with the Australian Strep A Vaccine Initiative (ASAVI) mentioned by one group as a collaborator on conducting a potential phase 2a clinical trial for their Strep A vaccine candidate.
The crucial role that CARB-X plays in the global AMR ecosystem was clear, with multiple presenters admitting that their projects and even their companies would simply not exist without the non-dilutive support provided to them by CARB-X.
AAMRNet recognises that connecting with the global AMR ecosystem is crucial to advancing Australian research and we are working to boost the chances of an Australian company presenting at such an important global event in the future.
To best enable this, Australia needs a dedicated AMR accelerator, and AAMRNet has been working to build the foundations for such an accelerator, one that can support local innovation and best prepare companies for applying to access the global funding pool provided by leading global organisations such as CARB-X.
It was a privilege to attend on the day, and our thanks goes to CARB-X for the kind invitation.
Hot Topics in AMR
Day One of ESCMID this year was designated an ‘Educational Day’ and featured interesting presentations and panel discussions on such topics as vaccine hesitancy and climate and health.
The day was highlighted by two afternoon sessions focused on translating the 2024 UNGA commitments on AMR into action. The sessions looked at using science to inform policy and several options that may help to achieve UNGA’s target of reducing deaths from AMR by 10 per cent by 2030.
The process behind the commitment to update the Global Action Plan on AMR was also outlined, with the first consultation set to open soon.
Another UNGA commitment, the establishment of an independent panel on evidence for action on AMR was also discussed and included a presentation describing learnings from the Intergovernmental Panel on Climate Change established in the late 1980s that could be applied.
While policy development can take time, AMR is killing millions of people around the world right now, and there are actions that can be taken now to reduce the impact of AMR. These include improving access to the newest antimicrobials. This is why AAMRNet has prioritised the urgent implementation of a subscription reimbursement mechanism in Australia, calling for Australia to support regional preparedness against AMR by improving access to effective antimicrobials in the South Pacific region. We will continue to advocate strongly on these priorities.
ATLAS Welcome Reception
The first day wrapped up at the ATLAS welcome reception, where it was great to catch up with new AAMRNet Steering Committee member Christine Carson. Supported by Pfizer, ATLAS (Antimicrobial Testing Leadership and Surveillance) is an interactive website that provides global antibiotic resistance surveillance data across multiple countries.
Day Two Starts with AAMRNet Member’s Keynote
The conference proper began on day two kicking off with the first keynote from AAMRNet Steering Committee member, Professor David Paterson, from the National University of Singapore. David spoke about the AMR situation in Asia, calling it the ‘hot zone’ of resistance.
Professor Paterson described the frightening mortality rates of carbapenem resistant Acinetobacter baumannii infections, with 50 per cent all-cause mortality rate at 30 days, a clear warning for any travellers thinking of hiring a moped on their trip to Bali!
Australia sits in the Western Pacific region, right on the doorstep of this ‘hot zone’, and he warned the room that what is happening in Asia today will be happening in other parts of the world, including Australia, tomorrow.
Professor Paterson did offer a glimmer of hope for the future, highlighting several new antimicrobials in the pipeline showing promise, including Indian companies supported by CARB-X, GangaGen and AAMRNet supporter Bugworks. These pipeline projects need to be supported and given the best chance of being commercialised and made available to patients.
Team Australia
It was great to see a strong Australian presence in Vienna for the congress. Steven Tong and Joshua Davis described outcomes from the Staphylococcus aureus network adaptive platform (SNAP) trial, and Professor Karin (Kas) Thursky from the Doherty Institute presented a keynote on ‘the new frontiers in antimicrobial stewardship’ on day three.
Other Australian representatives taking part included AAMRNet Steering Committee member David Grolman from Pfizer, as well as Australian Society of Antimicrobials (ASA) President A/Prof Louise Cooley, Alison Todd from SpeeDx, Jason Roberts from the Herston Infectious Diseases Institute (HeIDI) and Flavia Huygens from Brisbane-based diagnostic company, Microbio.
Poster Presentation Day
On Day three we presented our poster describing the extent of the unmet need in accessing antimicrobials in Australia. That unmet need is clear. On average, there were almost 500 applications per month from clinicians needing to access an unregistered antimicrobial in Australia, almost 30% of which were for treatment of a critically ill patient.
Pipeline Innovations Highlighted
Day four of the Congress, known as ‘Pipeline Monday’, highlighted innovations in infectious disease research from around the world. The scene was set by the World Health Organization’s Valeria Gigante, presenting the WHO’s analysis of the antibacterial and antifungal pipelines. Spoiler alert, its fragile!
Several other notable discussions focused on the potential of vaccines, both bacterial and viral, to combat AMR. One engaging session asked the question, ‘do we need new antibiotics?’, and the answer was a resounding yes!
Our poster was not the only one on display at ESCMID with links to AAMRNet, with Sohinee Sarkar from the Murdoch Children’s Institute presenting her research in an ePoster Flash Session on Pipeline Monday.
Sohinee presented on the results of a drug repurposing pipeline set up in Melbourne for resistant non-tuberculous mycobacteria. This project was supported in the early stages by a Therapeutic Innovation Australia (TIA) Pipeline Accelerator voucher awarded in the AMR specific round and delivered by AAMRNet in collaboration with TIA and CSIRO.
The ESCMID Global Congress is a huge event, with around 20,000 attendees and hundreds of presentations making planning essential in order not to miss a presentation that may be of interest.
AAMRNet was pleased to represent Australian efforts at such an important global event, presenting new research around the challenges of accessing antimicrobials in Australia, and continuing to build and strengthen global connections.
I am hopeful that in the next 12 months there will be further positive developments to showcase at the next global congress in Munich in 2026!