In 2023, almost one in five babies were admitted to a special care nursery or neonatal intensive care unit (NICU) in Australia, with about 20 per cent of those infants requiring respiratory support[1]. In the US, about 300,000 infants are admitted to NICUs annually[2].
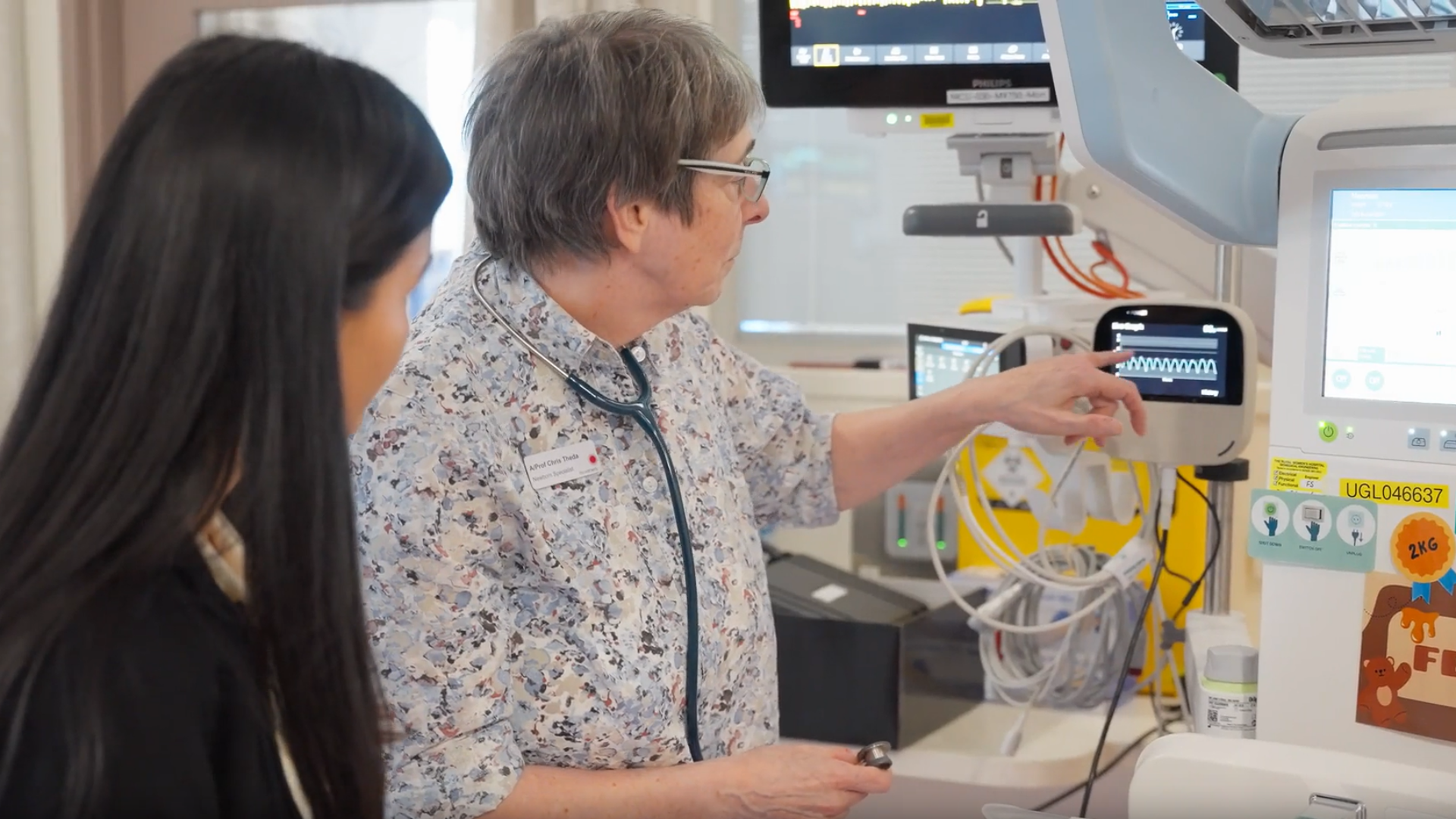
Newborns receive respiratory support through two main methods. The first method, mechanical ventilation, is highly invasive, requiring a tube inserted into the airway. For an infant, intubation carries extra risk, including airway trauma, lung injury, delayed oral feeding and neurological impairment. The second, and preferred method is non-invasive breathing support, such as Positive Airway Pressure (CPAP) or High-Flow Nasal Cannula (HFNC), , which uses a non-invasive nasal mask or prong interface.
For non-invasive methods, there is no way to accurately measure the pressure inside the airway in real time. This poses a problem as adequate pressure is needed to ensure that the treatment is effective and achieving good oxygen and carbon dioxide exchange, while too little or too much pressure could also cause injury to the babies’ fragile lungs. Even miniscule pressure errors can cause underventilation or lung injury, and the target pressure needs to be adjusted continuously based on the infant’s developing lungs and as their condition improves or worsens.
Recent studies have shown that improving the consistency of pressures delivered through CPAP leads to better clinical outcomes: less need for intubations, and less time on the ventilator[3],[4]. However, it comes with additional challenges: the interface can leak air, newborns often have obstructed nasal passages, and the required pressure may dissipate before reaching the airways. Importantly, the existing methods of optimising neonatal CPAP are resource-intensive, requiring one-on-one nursing care and manual support, and making it impractical for long-term or widespread adoption.
Seeking a better solution, Ventora Medical is transforming neonatal care with a world-first Airway Pressure Monitor that accurately monitors airway pressure to support breathing, reduce clinician uncertainty, enable tailored treatment, and improve clinical outcomes for newborns. This solution aims to reduce respiratory complications by decreasing NICUs’ reliance on mechanical ventilation. Ventora also expects its device to result in shorter hospital stays for families and significant cost savings for hospitals.
Leveraging existing clinical workflows
Ventora’s device is compatible with all non-invasive respiratory support systems, including CPAP and HFNC. This means the device has minimal impact on NICU workflows, reducing staff burden and lessening the potential for infection.
With $500,000 funding and support from MTPConnect’s Clinical Translation and Commercialisation Medtech (CTCM) program, Ventora Medical set out to develop and manufacture its clinical prototype and conduct a clinical feasibility study.
“Ventora is incredibly grateful to MTPConnect and its industry partners for supporting the development of Ventora’s clinical prototype,” said Ventora Medical Chief Executive Officer and Co-Founder, Mr Edward Buijs.
Compelling data propels progress
With wraparound support from MTPConnect during its CTCM project, including mentoring and guidance from their program partner Cicada Innovations, Ventora Medical progressed its technology from demonstrated safety in pre-clinical and one-hour clinical pilot studies to demonstrating safety and feasibility in its clinical feasibility study, marking a significant uplift in technology readiness.
Within two years, the team has delivered a clinical prototype and conducted a clinical feasibility study involving 22 patients receiving CPAP and/or HFNC respiratory treatments for an extended period between three and eight days.
Ventora Medical Chief Technology Officer, Co-Founder and Project Lead, Ms Amy Yu, explained the study successfully demonstrated technical and clinical proof of concept by using “the prototype device in a real-world clinical environment for a representative clinical-use duration”.
The team collected more than 2,000 hours of airway pressure data – combining this data with insights from a separate formative usability study to inform the next product design iteration including hardware, electronics, software, and algorithms.
The usability study, a CTCM project milestone, saw Ventora Medical partner with Sento Solutions over two days at the Epworth HealthCare Clinical Education and Simulation Centre, where neonatologists and neonatal nurses tested the prototype device in a representative clinical setting. The Airway Pressure Monitor received a System Usability Scale (SUS) score of 87, well above average, demonstrating strong user understanding of how to use the device safely and effectively.
Throughout the clinical trial, families were at the forefront of the team’s decision-making to ensure the device could function in a family-centred environment. Participant parents actively caring for their children, neonatal nurses, clinicians and respiratory therapists were all engaged in the design and study, providing feedback on how the device fit into or impacted their routines, which was incorporated into the final design.
The Ventora Medical team also received support in making key strategic decisions – determining which skills and capabilities to keep in-house and which to outsource. The team’s main learnings from the CTCM program were to engage manufacturers early, explore all options and remain flexible in design and manufacturing choices to reduce production costs without compromising performance or quality.
Ventora Medical has now completed the design of its Airway Pressure Monitor hardware, identified suitable manufacturing partners, and is preparing for transfer to manufacturing and pilot production.
Market positioning
While Ventora is looking to set a new gold standard in non-invasive ventilation for newborns, the economic benefits of its Airway Pressure Monitor are also compelling. Analysis conducted as part of the CTCM program projects that an average NICU in Australia could save $2–5 million annually through reduced mechanical ventilation use and shorter hospital stays.
Ventora’s intellectual property portfolio has also strengthened with the support of the CTCM program’s Value-Add Vouchers; providing an opportunity to conduct additional activities that complement the objectives of the CTCM project. Under this MTPConnect scheme, Ventora Medical was able to work on its IP strategy with FB Rice to coordinate and progress its existing patent applications. Three national-phase patents were granted for Australia, New Zealand and India, with applications in key jurisdictions the US, China and Europe still pending as of publication. This sets the scene for Ventora’s next objectives as it strategically positions itself in priority markets and explores potential manufacturing sites.
Looking beyond CTCM
The Ventora Medical team has worked steadily to attract $3 million in private investment and non-dilutive funding. Through an exciting new partnership with AusHealth, Ventora Medical expects to conduct a larger clinical study using its refined product. The timing of the Ventora Airway Pressure Monitor’s development aligns perfectly with growing global calls to use airway pressure monitoring in infants receiving non-invasive respiratory support.
In assessing the prototype performance in the clinical feasibility study, Ventora reassessed the prototype design, made adjustments and is now ready to proceed to production as part of its next steps beyond the CTCM program.
“The CTCM program has supported Ventora through a critical stage of development. It has accelerated the development of a clinical prototype and commencement of an ongoing clinical feasibility study,” said Ms Yu.
Ventora Medical’s proven technology and scalable business model, combined with the team’s dedication, openness to guidance and clear focus, position the company for future opportunities as it moves forward.
Discover more innovations in the Clinical Translation and Commercialisation Medtech Impact Report
________________________________________________________________________________________________________________________
[1] https://www.aihw.gov.au/reports/mothers-babies/australias-mothers-babies/contents/baby-outcomes/admission-to-a-special-care-nursery-or-neonatal-in
[2] https://www.cdc.gov/nchs/products/databriefs/db525.htm
[3] https://pubmed.ncbi.nlm.nih.gov/31572894/
[4] https://www.upmcphysicianresources.com/news/032719-bubble-cpap-and-preterm-neonates-a-noninvasive-ventilation-approach-to-quality-improvement