
Glaucoma is a group of eye diseases affecting more than 300,000 Australians, yet over half remain undiagnosed. This can lead to impaired vision and vision loss, reduced quality of life, a higher risk of injury and increased patient isolation and psychological distress1. Traditional treatments such as eye drops and laser therapy are often limited by poor patient compliance or may not be sufficient or suitable for everyone.
Surgically reducing intraocular (eye) pressure by allowing fluid to drain out of the eye is currently the most effective treatment. This includes procedures such as shunt placement, implant insertion and trabeculectomy. However, challenges persist around long-term efficacy, accessibility 2 3, complications4 and integration into routine care5. With patients typically requiring an average of 17 specialist follow-up visits in the first year and facing a 30–50 per cent failure rate within five years6 7,the need for more reliable and patient-friendly treatment solutions is increasingly urgent, especially for those in rural and remote regions.
Melbourne-based medical device company VividWhite is working to close these gaps and improve vision outcomes for people with glaucoma through its lead device, VividFlo (VW-51).
With CTCM funding, VividWhite has advanced VividFlo through an Australian multi-centre pivotal study and is producing promising data that address key challenges of current surgical treatments. It is hoped that the device will demonstrate improved reliability and a reduced post-surgery care burden, offering a significantly better treatment option to patients.
We’re grateful for the support of MTPConnect and the CTCM Grant program, which has been instrumental in advancing this work. As we move forward with local manufacturing and product registrations, our focus remains on delivering safe, effective, and accessible solutions for glaucoma patients.” – VividWhite Co-founder and CEO, Andrew Batty, said.
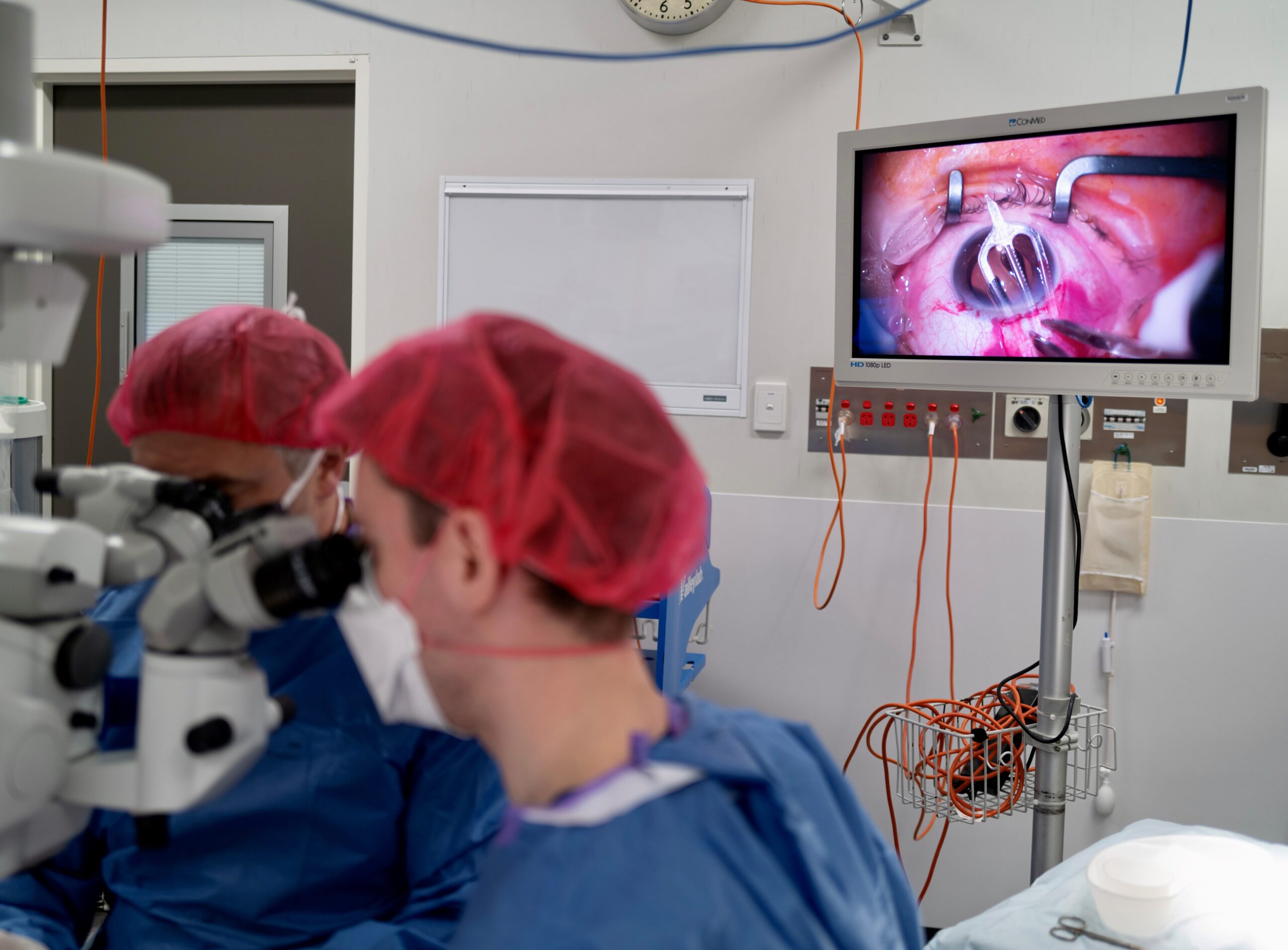
Surgical insertion of VividFlo in the eye.
Precision in pressure control
VividWhite’s device, the VividFlo glaucoma implant, is designed for surgical placement at the side of the eye, connecting to the anterior chamber to establish a sealed pathway that maintains stable intraocular pressure and enables precise fluid-flow regulation. Unlike conventional devices that rely on a single fluid channel, VividFlo features a network of resistive microfluidic channels with 157 points of egress. This device distributes fluid more evenly, alleviating fluid stress, safely reducing internal eye pressure.
Engineered with flexible materials and ultra-small dimensions, VividFlo prioritises patient comfort and improves ease of use for surgeons. Its implantation technique is intentionally designed to be intuitive, closely mirroring existing ‘tube and plate’ procedures, making it easy for trained ophthalmologists to adopt.
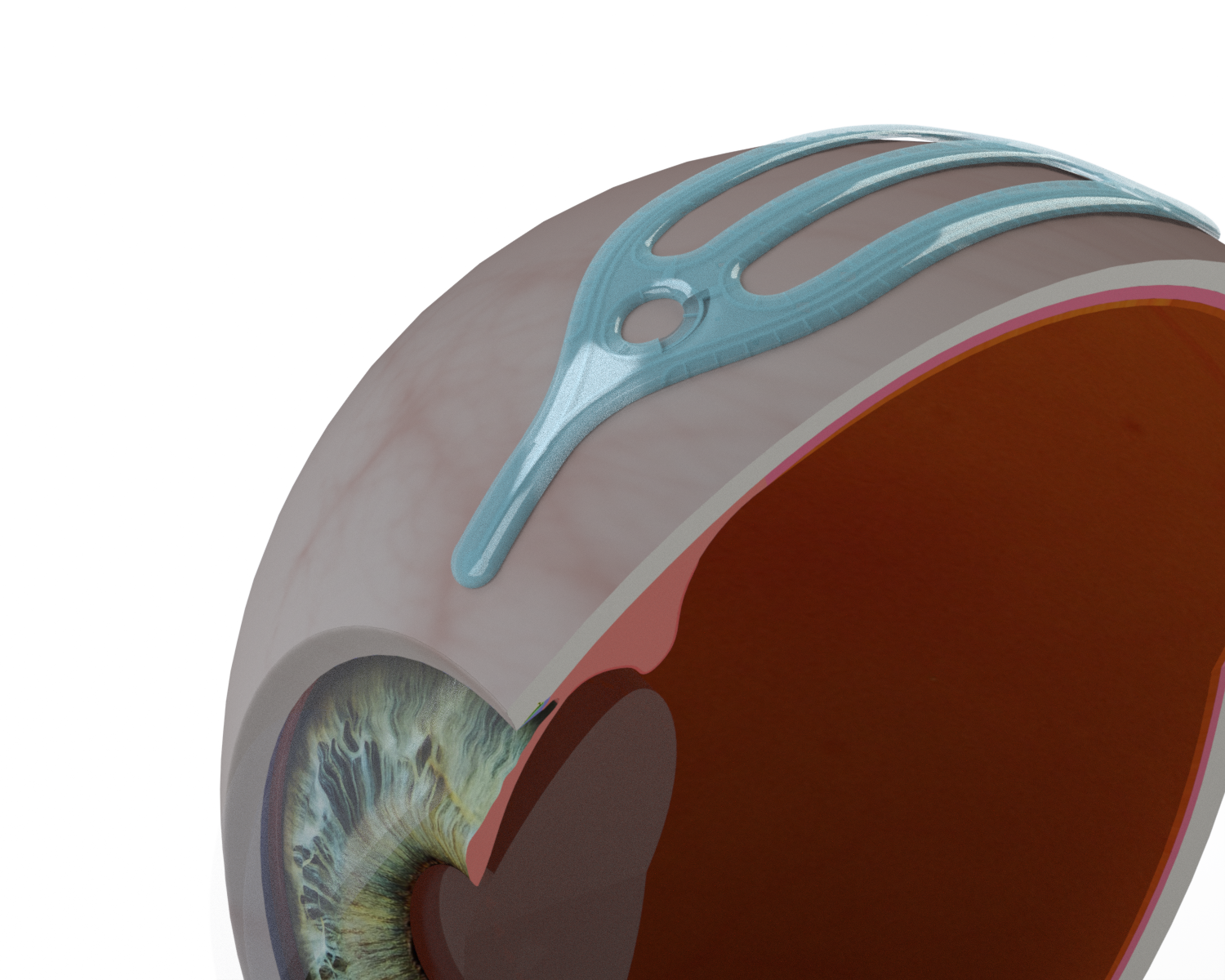
Positioning VividFlo in the eye to relieve intraocular pressure.
Overcoming barriers to clinical trial recruitment
When VividWhite joined the CTCM program, it had already achieved encouraging clinical results in efficacy and reduced post-operative care from a feasibility study involving 10 participants. The study also provided manufacturing insights that informed the product’s design and the execution of the larger pivotal trial.
While the journey presented challenges, the team adapted quickly, supported throughout by MTPConnect and its program partner, the Medical Device Partnering Program (MDPP). The biggest hurdle during the two-year project was recruiting participants for the pivotal study, which progressed slower than expected.
In response, the team swiftly expanded the trial to additional sites, to eventually include 6 clinical and 7 surgical sites, with 10 active clinicians involved in the study. This paved the way for more investigational teams to integrate the new treatment into existing clinical workflows. Although this required extensive communication and training, it provided valuable insights into the device. Throughout the ongoing pivotal trial, investigators have worked closely with the VividWhite team, providing insights into the device, its
implantation and post-operative management requirements. Their feedback – particularly on surgical challenges and how best to convey key differences in handling between VividFlo and existing competitor devices – has been instrumental in developing the product dossier.
By expanding test sites, increasing the number of investigator teams and broadening testing parameters, VividWhite successfully recruited 65 participants – five more than originally planned – and, at the time of publishing, achieved promising trial outcomes. In overcoming this major hurdle, VividWhite’s carefully tailored strategy and strong relationships with all investigational teams have paid off, laying a solid foundation for future trials.
“With the completion of recruitment for VividWhite’s pivotal glaucoma trial, we’ve reached an important milestone in our efforts to improve vision care,” VividWhite Co-founder and CMO, Professor Michael Coote, said.
Adding value on manufacturing, trial execution and regulatory strategy
As part of its strategic planning, VividWhite leveraged the CTCM’s Value-Add Voucher scheme with infrastructure partner Therapeutic Innovation Australia to unlock targeted support ahead of its pivotal trial. This included engaging the Victorian node of the Australian National Fabrication Facility (ANFF), the Melbourne Centre for Nanofabrication, to produce specialised tooling for implant manufacturing. This allowed VividWhite’s engineering team to deepen their expertise in commercial-scale production. These learnings have positioned the company to expand local manufacturing capabilities in future phases.
Further voucher support was directed towards clinical trial oversight, regulatory compliance, and clinical trial data integrity, delivered in partnership with Avania. This input directly shaped the structure and execution of the pivotal trial. A final collaborative activity with Commercial Eyes, facilitated through the CTCM program, helped strengthen VividWhite’s regulatory strategy to enter the US market.

Getting ready for market
VividWhite successfully progressed its patent application to the national phase, securing review across key global jurisdictions including Australia, China, USA, EU, Canada and Japan. In addition, regulatory activity will reflect the market opportunity and alignment with the company’s commercial road map.
As VividWhite continues to monitor patient safety, its fundraising efforts are now geared towards completing the pivotal trial, publishing the results and preparing to scale manufacturing. Ongoing engagement with the local community of ophthalmologists – particularly through presenting clinical study data at the Australian and New Zealand Glaucoma Society’s 2024, 2025 and 2026 congress – is helping to keep VividFlo visible within the clinical community and is critical to supporting its transition from clinical validation to commercial readiness.
“It’s quite a privilege to be part of this trial, based on such fantastic research and a design that has been entirely locally created,” said Professor Keith Martin, Managing Director at the Centre for Eye Research Australia (CERA) and one of the surgeons participating in the study. “There are not many cases where a small team, without assistance from a major medical device or pharmaceutical company, has taken an idea like this all the way from the drawing board to clinical trial”.
Recent funding milestones include a Gate 2 pre-seed investment from ANFF-C to support manufacturing and packaging development, and a LaunchVic Medtech Market Growth Program grant to advance VividWhite’s market expansion plans.
Together, these grants reflect growing momentum and validate the team’s strategic execution across clinical and regulatory domains, signalling strong progress towards the company’s next milestones in bringing this new implant to patients to save their vision.
1. https://glaucoma.org.au/news-details/treatment/glaucoma-update-2024-the-treatment-landscape
2. https://pubmed.ncbi.nlm.nih.gov/22814041/
3. https://pubmed.ncbi.nlm.nih.gov/26664740/
4. https://www.aoa.org/assets/documents/EBO/931-016GEDDE-2007.pdf
5. https://pubmed.ncbi.nlm.nih.gov/25999687/
6. https://pubmed.ncbi.nlm.nih.gov/34715397/
7. https://onlinelibrary.wiley.com/doi/10.1155/2015/847439